Key points
Congenital absence or loss of the penis is a devastating condition.
Gender reassignment is no longer an option for this condition.
Phalloplasty is the gold standard treatment for this condition.
Maximal conservation of any penile tissue and incorporation in the phallus must be considered.
Phalloplasty must be performed by experienced surgeons.
Complication rate of phalloplasty is high.
Erectile implants after phalloplasty are feasible.
Introduction
The biological male without a penis or with an insufficient penis remains a major challenge. Failure of penile development, trauma, medically-indicated penile amputations, and failed reconstructions of congenital anomalies are the main reasons for penile insufficiency(Table 1). Severe penile insufficiency and absence of a penis are devastating conditions for men, with significant psychological and physical impact. Although uncommon, it is a challenging condition to treat.
Table 1: Conditions leading to severe penile insufficiency. |
Congenital conditions (disorders of sexual development) |
Aphallia or penile agenesis
Idiopathicmicropenis
46 XY DSD
Exstrophy
Cloacalexstrophy |
Genital trauma |
Injuries
Surgery |
Penile amputation |
Treatment options for this devastating condition are gender reassignment, tailoring of the penile stump, penile reattachment, phallic reconstruction (phalloplasty), and most recently penile transplantation (Table 2).
In the past, sex reassignment to the female gender had been offered based on the principles applied to newborns with disorders of sexual differentiation (DSD) and ambiguous genitalia. There is no evidence to demonstrate that the outcome of this policy is satisfactory. Indeed, long-term evaluation of a few patients shows contradictory results, which have triggered great controversy of this therapy [1]. Some recent reports have alerted physicians to the high incidence of gender identity disorder in gender reassigned children. Especially in cloacalexstrophy, the results have been disappointing [2,3]. Today, sex reassignment is no longer considered treatment of choice. Parents of children presenting with these conditions should be informed that a phalloplasty can be performedlater in life avoiding the stressful decision of gender reassignment.
Table 2Treatment options for severe penile inadequacy. |
Endocrinological treatment
Penile reconstruction
Penile replacement (phalloplasty)
Gender reassignment
“Penile transplantation”
“Tissue engineering” |
Tailoring of the penile stump by means of penile degloving, division of the suspensory ligament, and rotational skin flaps has been reported [4,5]. However, this can only be applied to moderate penile injuries where there is still a reasonable penile stump.
When penile skin is lacking for reconstruction a reversed mesh graft can be used to cover the penile shaft. Widely meshed split-thickness skin grafts should never be used on the penile shaft. However, with the so‐called ìrreversed mesh graft (i.e. nonexpanded or 1:1 meshed skin grafts) only puncture‐type perforations are made with a V1 carrier (Humeca Ltd., Enschede, the Netherlands). These allow for a better fluid evacuation and thus a better skin graft take whilst still avoiding poor scarring and an unaesthetic mesh pattern in the long-term. These nonexpanded meshed split‐thickness grafts have also become a mainstay of complicated, staged urethral reconstruction [6,7]. McAninch [8] has described the use of (not too widely) meshed split-thickness skin grafts for scrotal reconstruction. Meshed grafts in this location may produce good esthetic results, but they will never measure up to the functional capacity of sheet or 1:1 perforated grafts.
In some cases good results can be obtained by combining techniques (Figure 1).
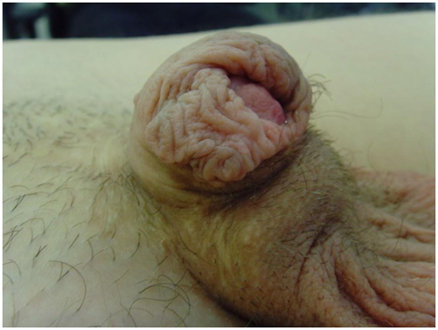
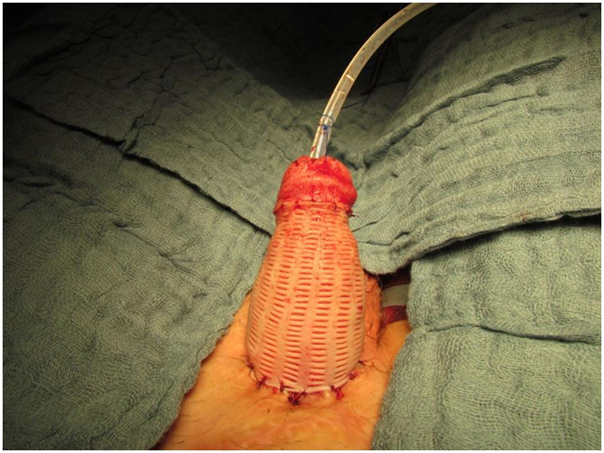
Figure 1:Example of a cripple penis where the stump was released and covered with reversed mesh graft
Penile reattachment can be attempted in the acute phase following traumatic amputation of the penis. The survival of the reattached penis depends on the viability of the amputated segment and the condition of the graft bed or penile stump. Reattachment must take place within 24 hours. Current reattachment techniques rely on microsurgical approximation of the dorsal structures and cavernosal arteries with uniformly good results. In traumatic amputation, salvage of the amputated segment with reattachment is the primary treatment option [9]. The outcome of erectile function after reattachment is, however, not clear.
The ideal candidate for genital replantation is a patient with a clean, sharp cut in which the amputated part has been cooled.
Recently, two case report has been published on penile transplantation, one unsuccessful [10] and more recently a successful case . This technique is still experimental and is not a current treatment option. Future options, like tissue engineering, are as yet matters for science fiction rather than science.
Phallic reconstruction is another treatment option. The first phallic reconstruction was described by Bogoras in 1936 with a tubed abdominal flap [11]. Phalloplasty procedures have followed the evolution and advances made in plastic surgery. Originally, it was a complex, time-consuming, multistage procedure using tubed skin flaps or pedicledmyocutaneous flaps with variable and suboptimal results [12-14].
In 1984, Chang completed the first successful microsurgical phalloplasty with a radial forearm free flap [15]. Since then, the radial forearm flap has been widely accepted as the best donor site for penile reconstruction and is nowadays the gold standard in penile replacement for female-to‐male transsexuals [16–18]. This technique can also be applied to boys without an adequate penis. Defining penile insufficiency is difficult. To base the definition of inadequacy on length and appearance alone is impossible, especially in infants and young children. Penile inadequacy is an individual diagnosis that can only be made after puberty when sexual development is completed and the patient is sexually active. Puberty can change the final outcome of penile length and girth substantially. The number of children diagnosed with micropenis persisting into adulthood is limited [19].
As for penile reconstruction, many techniques have been described for penile augmentation. However, the results of most of these surgeries are very limited, Indeed, the reported outcome is often poor [20].
In this chapter, we will focus on the phalloplasty. It is considered the gold standard for penile absence or severe penile inadequacy, when endocrinological therapy is not beneficial.
Phalloplasty
Surgical reconstruction of the penis (phalloplasty) is difficult because of the different cosmetic and functional requirements of the patients:
1. The reconstructed penis should be esthetically acceptable; it must be as normal as possible in appearance (minimal scar, glandular reconstruction, etc.).
2. The penile shaft must contain a urethra that extends to the distal tip and permits the patient to void in a standing position unless there is a concomitant condition that makes normal voiding impossible.
3. The penile shaft should allow the implantation of a penile stiffener in order to regain the possibility of sexual intercourse. Therefore, protective and erogenous sensation is needed.
4. The donor area should cause minimal morbidity with an acceptable scar that is easy to conceal.
Many of these objectives could not be obtained with the older methods in which phallic reconstruction required a complex multistaged procedure. Nowadays, microvascular free flap techniques come closer to achieving these objectives. Despite the multitude of free flaps that have been published (frequently as a case report), the radial forearm free flap is universally considered the gold standard in penile reconstruction [16–18,21].
Surgical technique
At Ghent University Hospital, more than 500 consecutive patients have undergone phallic reconstruction using a radial forearm flap. This experience is mainly in female-to-male transsexuals. We describe our current technique as well as the different changes and refinements madein the reconstruction for the biological male in this radial forearm phalloplasty procedure.
Depending on the underlying condition, we try to preserve any useful penile and cavernosal tissue. The urethral stump, if available, is prepared for connection with the phallic urethra and, also if available, a dorsal penile nerve is identified. A free vascularized flap of the forearm and the creation of a phallus with a tube-in‐a‐tube technique is performed with the flap still attached to the forearm by its vascular pedicle. A small skin flap and skin graft are used to create a corona and imitate the glans of the penis.
The free flap is then transferred to the pubic area where first the urethral anastomosis is performed. The radial artery is then microsurgically connected to the common femoral artery in an end-to‐side fashion, usually with an interpositional vein graft taken at the ankle. The venous anastomosis is performed between the cephalic vein and the greater saphenous vein. One forearm nerve is connected to the ilioinguinal nerve for protective sensation, and the other nerve is anastomosed to one of the dorsal penile nerves for erogenous sensation. All patients receive a suprapubic urinary diversion postoperatively.
In the first 50 patients the defect on the forearm was covered with full-thickness skin grafts taken from the groin area; later, patients were given split‐thickness skin grafts harvested from the medial and anterior thigh.
The patients remain in bed for one week after which the transurethral catheter is removed. Three to five days later the suprapubic catheter is clamped and voiding is started. It sometimes takes several days before good voiding is observed. The average admission period for the phalloplasty procedure is approximately 2.5 weeks.
Tattooing of the glans can be performed after a 2–3 month period, before sensation returns to the penis (Figure 27.4).
The development of perforator flaps has given rise to some new reconstructive options in patients with severe (congenital) penile insufficiency. Perforator flaps have the advantage of reducing the donor site morbidity, increasing the range of motion and combining different tissue flaps on one single pedicle. Although – overall – the free vascularized radial forearm flap described above is still considered as the “standard technique” in penile reconstruction, the pedicledantero‐lateral thigh (ALT) flap has been shown to provide a valuable phalloplasty alternative specifically in patients with congenital penile insufficiency. This flap is a skin flap based on a perforator from the descending branch of the lateral circumflex femoral artery, which is a branch from the femoral artery. There are several reasons why in boys without a penis a pedicled ALT flap can be preferred above the standard radial forearm flap :
a pedicled flap reconstruction (the flap has a sufficiently long pedicle) avoids the technically more complex microscopic procedure and might also shorten the operation time
A visible donor site scar on the forearm, often considered as the signature of female-to‐male transsexualism, is avoided and the donor site on the leg can more easily be concealed.
Previous reconstructive surgeries at the pelvis, groin area and lower abdomen (e.g. in case of bladder exstrophy) might have altered the local anatomy and vasculature making a microsurgical anastomosis more difficult.
The subcutaneous fat layer is much thinner than in a (biologically female) trans-man, facilitating the (urethral)tube-within‐a‐(penile)tube reconstruction of the penis; moreover, many exstrophy patients empty their bladder by catheterization through a continent diversion (e.g. appendico‐vesicostomy) and don't require a urethral reconstruction in their phalloplasty. An ejaculatory opening can be left at the ventral aspect just above the scrotum.
It is very important to preserve and incorporate any useful glandular, penile and cavernosal tissue at the basis of the newly-reconstructed phallus in order to facilitate sexual stimulation and pleasure (Figure 27.5d). If available, a dorsal penile nerve is identified and connected with a cutaneous nerve of the flap; if not available, the lateral femoral cutaneous nerve is connected to the ilioinguinal nerve.
Usually, it is recommended to perform a 3D angio CT-scan preoperatively to provide detailed information on the perforator vessel(s) and the subcutaneous tissue layer.
Sexual function
Sexual function and pleasure is one of the goals in phallic reconstruction. For this purpose, in biological males any sensitive penile tissue left must be incorporated. Any glans tissue present can be incorporated in the base of the neophallus. This is important for sexual stimulation and pleasure. Further erogenous and tactile sensations of the neophallus are obtained by microscopic anastomosis of, respectively, one dorsal phallic nerve and one ilioinguinal nerve to the cutaneous nerves of the flap [22].
Obtaining sufficient rigidity of the penis to allow penetration is extremely difficult because there is no good substitute for the unique erectile tissue of the penis. The radial forearm flap is too soft and can even demonstrate some atrophy of the subcutaneous fat with a loss of more than 20% of circumference. The use of bone or cartilage grafts has often resulted in complications and failure because of resorption, curving, or fracture [23–24]. For sexual penetration, a penile stiffener is needed and, fortunately, the radial forearm flap has sufficient subcutaneous bulk to permit incorporation of a penile prosthesis. Incorporation of a penile stiffener can only be done after the phallus is endowed with sufficient protective sensation, which usually takes at least 12 months. Good protective sensation is critical in preventing breakdown and erosion of an internal stiffener [25–26]. Next to sensitivity, urethral function also has to be considered. Implantation of a penile prosthesis must be withheld until the urethra is stable, and the patient is free of voiding symptoms and urinary tract infection [25].
Unfortunately, high erosion rates (20–50%) are reported [16,25]. One of the reasons could be the less vascularized skin and subcutaneous tissue of the neophallus (in comparison with a native impotent penis), which can lead to chronic ischemia after implantation of a stiffener and subsequently diminished resistance against infection and perforation.
Despite the complications and difficulties, the satisfaction rate after phalloplasty is high and the results cosmetically pleasing (Figure 1). None of our patients regret the surgery. An important boost concerning the self-esteem level is observed in each patient, which is a very important outcome factor postoperatively.
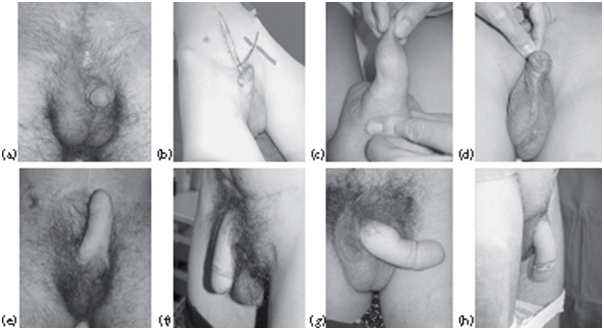
Figure 1Results of phalloplasties in biological males. (a) and (a) Cripple exstrophy.(c)Epitheloied sarcoma of penis.(d)Cripple hypospadias with absence of corpora). (a)–(d) Before surgery; (e)–(h) after surgery.
Complications
Despite the good outcomes described, phalloplasty is associated with a high complication rate. Most complications are related to the urethral reconstruction [16,27–28]. The main complications are fistulae and stenoses whenever the urinary tract is attached to the native urethra. Despite the high prevalence of these complications, the literature on treatment of these complications is sparse. Consequently, always consider whether the urethral reconstruction is necessary, as some boys will not need urethral reconstruction as the bladder is augmented and diverted. In addition, it is important to consider the ejaculation of the patient.
Is he actually ejaculating? Where? If so, do we want to reconstruct the urethra for ejaculation alone, or do we want to keep the ejaculation where it is? Preference should be given to keep the ejaculation where it is as reconstructing a urethra just for ejaculation can result in higher complications and possible anejaculation due to the length of the urethra and the weakness of the ejaculation due to the underlying condition.
Urethral fistulas
Remove all scar tissue and try to bring well-vascularized tissue to the area that needs reconstruction. In radial forearm flap phalloplasty, we have to consider that the tissue of the urethra is skin and not mucosa. Larger stitches with cutting needles should be used. The duration of bladder drainage is unknown, but in our practice we prefer to drain for 12 days. When using local skin flaps, consider possible future hair growth in the urethra. A good alternative is the use of buccalmucosa[29].
Urethral stenosis
In our experience we first attempt an endoscopic incision of the stenosis, if the stricture is relatively short. We leave a catheter for 12 days, which is much longer than after urethrotomy for urethral stenosis in normal urethras. We have to remember that part of the urethra is composed of skin, and healing of skin lesions is much slower than mucosal healing.
If endoscopic incision fails, we perform a formal urethroplasty. End-to‐end anastomosis or Heineke‐Mikulicz type reconstructions with longitudinal incision over the stenotic area closed transversally. For longer strictures or complex and repetitive stricture, 2‐stage urethroplasty (Johansson) must be considered. The stenotic area is longitudinally incised and the borders of the stricture are sutured to the surrounding healthy skin. The urethra remains opened until the skin and urethra are well healed, which usually takes a minimum of three months. During the second stage of the urethroplasty, the urethra is closed again and covered with skin.
Experience with phalloplasty in biological males
There are only a few series published on this topic. Perovic reported phalloplasty in 24 patients without a functional penis using the extended pedicle island groin flap. He suggests this technique as an available alternative to the microsurgical free tissue phalloplasty [30].
Sengezeret al. suggested total penile reconstruction with sensate osteocutaneous free fibula flap. With this technique, promising results were obtained in 18 patients without a functional penis for different reasons [31]. Gilbert et al. were the first to describe the application of a radial free forearm flap for phallic reconstruction in 11 boys without a functional penis. Satisfactory results were obtained [32].
We performed phalloplasty in 19 males with the use of the radial forearm flap: eight boys with inadequate penis after exstrophy repair; one boy with penile loss after multiple hypospadias repair; six boys with DSD andmicropenis; one boy with a penile epithelioid sarcoma; one boy with ideopathicmicropenis; and two men who traumatically lost their penis. There were no complications concerning the flap. Two complications were reported in the early postoperative period: one pulmonary embolism and one severe hematuria. Four patients developed urinary complications (stricture and/or fistula) for which a secondary procedure was necessary.
In a further five boys –two with cripple exstrophy and three after cloacalexstrophy – phalloplasty was done with an ALT flap..
Patient satisfaction after surgery was high in 22 cases and moderate in two cases. Psychological evaluation confirms this, especially on the self-esteem level. Ten patients underwent erectile implant surgery. In two patients, the erectile implant had to be removed because of infection (unpublished data).
In our series, one adolescent patient presented with epithelioid sarcoma of the penis and the phalloplasty was performed in a 1-stage procedure with the penectomy [33]. This could be an option for some patients undergoing penile amputation if oncologically acceptable.
The reported success of phalloplasty in boys without a functional penis has convinced us that penile reconstruction is the optimal treatment for this condition. It has extremely good results and improves their self-esteem and their physical and psychological wellbeing. But the complication rate of the erectile implants is high.
Phalloplasty opens new horizons for the treatment of penile agenesis, micropenis, crippled penis, shrivelled penis, some disorders of sexual development (DSD) conditions, traumatic amputations in which the amputated segment is lost for replantation, iatrogenic amputations, and cloacalexstrophy.
References
1. Woodhouse CR. Sexual function in boys born with exstrophy, myelomeningocele, and micropenis.Urology1998;52:3–11.
2. Reiner WG, Gearhart JP. Discordant sexual identity in some genetic males with cloacalexstrophy assigned to female sex at birth.N Engl J Med2004;350:333–41.
3. Mayer-Bahlburg HF. Gender identity outcome in female-raised 46, XY persons with penile agenesis, cloacalexstrophy of the bladder, or penile ablation.Arch Sex Behav2005;34:423–38.
4. Ochoa B. Trauma of the external genitalia in children: amputation of the penis and emasculation. J Urol1998;160:1116–19.
5. Amukele SA, Gene WL, Stock JA, Hanna MK. 20-year experience with iatrogenic penile injury.J Urol2003;170:1691–4.
6. Schreiter F, Noll F.Mesh graft urethroplasty. WorldJ Urol1987;5:41.
7. Schreiter F, Noll F, Mesh. Graft urethroplasty using split thickness skin graft for foreskin.J. Urol1989;142:1223.
8. McAninch JW. Management of genital skin loss.UrolClinNorth Am1989;16:387.
9. Jezior JR, Brady JD, Schlossberg SM. Management of penile amputation injuries.World J Surg2001;25:1602–9.
10. Hu W, Lu J, Zhang L, Wu W, Nie H, Zhu Y, et al. A preliminary report of penile transplantation.Eur Urol 2006;50:851–3.
11. Bogoras NA. Uber die volle plastische wiederherstellung eines zum koitus fahigen penis (Penisplastica totalis). ZentralblChir1936;22:1271.
12. Hoopes JE. Surgical reconstruction of the male external genitalia.ClinPlastSurg1974;1:325.
13. Orticochea M. A new method of total reconstruction of the penis.Br J PlastSurg1972;25:347.
14. Puckett CL, Montie JE. Construction of male genitalia in the transsexual, using a tube groin flap for the penis and a hydraulic inflation device.PlastReconstrSurg1978;61:523–30.
15. Chang TS, Hwang WY. Forearm flap in one stage reconstruction of the penis.Plast Reconstr Surg 1984;75:251.
16. Monstrey S, Hoebeke P, Dhont M, Selvaggi G, Hamdi M, Van Landuyt K, et al. Radial forearm phalloplasty: a review of 81 cases.Eur J PlastSurg2005;28:206–12.
17. Gilbert DA, Horton CE, Terzis JK, Devine CJ, Winslow BH, Devine PC. New concept in phallic reconstruction.Ann PlastSurg1987;18:128.
18. Hage JJ, Bloem JJ, Suliman HM. Review of the literature on techniques for phalloplasty with emphasis on the applicability in female-to‐male transsexuals.J Urol1993;150:1093–8.
19. Lee PA, Houk CP. Outcome studies among men with micropenis.J PediatrEndocrinolMetab2004;17:1043–53.
20. Li CY, Kayes O, Kell PD, Christopher N, Minhas S, Ralph DJ. Penile suspensory ligament division for penile augmentation: indications and results.EurUrol2006; 49:729–33.
21. Gottlieb LJ, Levine LA.A new design for the radial forearm free-flap phallic reconstruction.Plast Reconstr Surg 1993;92:276–84.
22. De Cuypere G, T'Sjoen G, Beerten R, Selvaggi G, De Sutter P, Hoebeke P, et al. Sexual and physical health after sex reassignment surgery.Arch Sex Behav2005;34:679–90.
23. Ali M. Surgical treatment of the male genitalia with special reference to the use of periosteal bone graft in constructing the penis.J IntCollSurg1957;27:352.
24. Khouri RK, Young VL, Casoli VM. Long-term results of total penile reconstruction with a prefabricated lateral arm free flap.J Urol1998;160:383–8.
25. Jordan GH, Alter GJ, Gilbert DA, Horton CE, Devine CJ. Penile prosthesis implantation in total phalloplasty.J Urol1994;152:410–14.
26. Hoebeke P, Decuypere G, Ceulemans P, Monstrey S. Obtaining rigidity in total phalloplasty: experience with 35 patients.J Urol2003;169:221–3.
27. Hage JJ, Bloem JJ. Review of the literature on construction of a neourethra in female-to‐male transsexuals.Ann PlastSurg1993;30:278–86.
28. Hoebeke P, Selvaggi G, Ceulemans P, De Cuypere G, T'Sjoen G, Weyers ,Set al. Impact of sex reassignment surgery on lower urinary tract function.EurUrol2005;47:398–402.
29. Rohrmann D, Jakse G. Urethroplasty in female-to‐male transsexuals.EurUrol2003;44:611–4.
30. Perovic S. Phalloplasty in children and adolescents using the extended pedicle island groin flap.J Urol1995;154:848–53.
31. Sengezer M, Oztürk S, Deveci M, OdabaÁi Z. Long-term follow‐up of total penile reconstruction with sensate osteocutaneous free fibula flap in 18 biological male patients.PlastReconstrSurg2004;114:439–50.
32. Gilbert DA, Jordan GH, Devine CJ, Winslow BH, Schlossberg SM. Phallic construction in prepubertal and adolescent boys. J Urol 1993;149:1521–6.
33. Hoebeke PB, Rottey S, Van Heddeghem N, Villeirs G, Pauwels P, Schrauwen W, et al. One-stage penectomy and phalloplasty for epithelioid sarcoma of the penis in an adolescent.EurUrol2006;51(5):1429–32.












